Post#3: Hyperbaric Oxygen Therapy (HBOT) is the De Facto Treatment for Brain Injuries. Deal With It.
Disclaimer: I am not a doctor...blah, blah, blah. Yet, the data stubbornly keeps pointing out the obvious: HBOT has shown consistent healing effects for traumatic brain injury/neurological injury.
HBOT Coming to the Forefront
Well, well, well…look who beat me to the punch: The hardest working scientist in the public sphere, the Popular Rationalist himself Dr. James Lyons-Weiler interviewed Dr. Paul Harch, M.D. The presentation focused on COVID infection treatment, Long-Haul COVID and Vaccine Injury. This was jam-packed with real-information and real science, not the lame CDC/FDA propaganda.
I am happy and relieved that there is now a convergence from so many different sources on the incredible power of HBOT to treat a myriad of different conditions, including COVID19. But HBOT is increasingly being applied to neurological conditions and other injuries.
Paul Harch recently published a comprehensive review and meta-analysis of the current state-of-the-art in HBOT for mild traumatic brain injury (mTBI) in Frontiers of Neurology. His review of the clinical trials and case reports clearly and systematically demonstrated that the evidence is at Class 1 level (the highest) under Centre for Evidence-Based Medicine and Class A (highest) Practice Recommendation under the American Society of Plastic Surgeons Grade Practice Recommendations for the use of hyperbaric oxygen at 1.5 ATA for mTBI/PPCS. Medicare, Medicaid, AMA, APA, and any insurance companies are on notice that HBOT IS Standard of Care (SOC) for mTBI/PPCS.
This is in agreement and supports the best-evidence that was available at the time Jim Wright and I wrote our review in the journal Neurology (2016), where we presented the clinical evidence at that point in time:
With Dr. Harch’s publication, we now have an up-to-date review (by an expert) that HBOT is AWESOME in treating mTBI/PCS/PPCS. But I also have my own analysis.
Hyperbaric oxygen: A-level evidence in traumatic brain injury clinical trials
In the hyperbaric medicine literature, in the last 16 years, has provided a set of studies of sufficient rigor and standardized methodology for analysis in the mild-to-moderate TBI/ post-concussion syndrome/persistent post-concussion syndrome (mTBI/PCS/PPCS). The heterogeneity of a brain injury makes it difficult to diagnose or to even agree on the classification and extent of damage and requires a complex coordination and treatment approach for some form of recovery. The notion that the central nervous system can be repaired and regenerated is a relatively new concept in medicine. Prior to the 1990’s, the idea that new neurons can be re-generated in adulthood was considered a dubious proposition [1, 2]. Now, with more basic and clinical research, we know that it is possible to regenerate CNS tissue and repair is possible [3-5].
Clinical trials in hyperbaric oxygen therapy (HBOT) nationally and internationally has provided a set of clinical trials and case reports that can be analyzed and compared across studies (Table 1) [6-23]. Symptoms scores, as well as computerized neurocognitive task assessments, have been used across multiple studies that allow a relative assessment and comparison. In some cases, the use of imaging (such as SPECT and MRI) have provided much needed information as to changes in blood flow in cerebral regions and changes in tract density [10-12, 16, 18, 19, 22]. All studies report a reduction in symptoms and an increase in quality of life for those that have undergone HBOT and the presumed inert control group (hyperbaric air).
Table 1 – English language case reports and clinical trials using hyperbaric oxygen in patients diagnosed with a traumatic brain injury (TBI). All injuries are in the mild TBI category unless noted. Statistically significant improvements (a=0.05; P < 0.05) are denoted by a Yes designation for the tests that were applied to assess symptoms and neurocognitive performance as the difference between baseline measures and assessment after the last hyperbaric oxygen therapy treatment. N/A, not applicable. SOC, standard of care. RCT, randomized-controlled clinical trial. Retro, retrospective clinical analysis. Pre to Post, trial design analyzed the difference between the patients starting (baseline) assessment and after finishing the last hyperbaric oxygen treatment. Level of Evidence, classification of studies as I-IV as defined by the American Academy of Neurology guidelines.
Technical Difficulties: Sham and Placebo Controls
HBOT is a combination therapy that requires both pressure + gas (usually as Oxygen at ≥ 99% purity) and time at depth. The Undersea & Hyperbaric Medical Society (UHMS) definition for hyperbaric oxygen therapy (below) requires a pressure of > 1.4 ATA in order to elicit a “clinically therapeutic” effect. Of note, the definition that UHMS (website: https://www.uhms.org/resources/hbo-indications.html) uses for HBOT is the following:
“The UHMS defines hyperbaric oxygen (HBO2) as an intervention in which an individual breathes near 100% oxygen intermittently while inside a hyperbaric chamber that is pressurized to greater than sea level pressure (1 atmosphere absolute, or ATA). For clinical purposes, the pressure must equal or exceed 1.4 ATA while breathing near 100% oxygen. The United States Pharmacopoeia (USP) and Compressed Gas Association (CGA) Grade A specify medical grade oxygen to be not less than 99.0% by volume, and the National Fire Protection Association specifies USP medical grade oxygen…According to the UHMS definition and the determination of The Centers for Medicare and Medicaid Services (CMS) and other third party carriers, breathing medical grade 100% oxygen at 1 atmosphere of pressure or exposing isolated parts of the body to 100% oxygen does not constitute HBO2 therapy. The patient must receive the oxygen by inhalation within a pressurized chamber. Current information indicates that pressurization should be to 1.4 ATA or higher.”
The definition of HBO therapy is a policy recommendation. The cut-off for declaring a treatment pressure and gas mixture as therapeutic is an empirical determination that is tailored to the conditions and organ systems that respond to those treatment parameters. The use of air pressure groups as sham or placebo controls is not supported by the peer-reviewed literature for neurological injury studies. This needs to be repeated for emphasis: there is no peer-reviewed evidence that pressurized air can be used as a placebo or sham for neurological injury studies in hyperbaric oxygen therapy research. Indeed, an excellent summary review of the hyperbaric literature conclusively makes the case that pressurized air cannot be a sham or placebo [24] and should be viewed as having reparative effects. Until a dosing study is carried out to determine the minimum pressure + Oxygen concentration and time at depth that will NOT elicit a response, the use of a pressurized air group should not be considered as a valid control comparator.
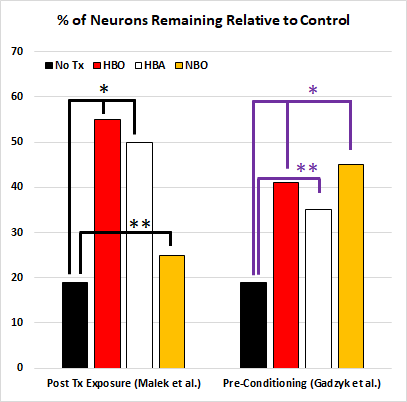
Animal studies carried out by Malek et al [25] and Gamdzyk et al [26] using hyperbaric air (HBA), hyperbaric oxygen (HBO) and normobaric oxygen (NBO; 100% oxygen at ambient pressure) make this point abundantly clear. Induction of forebrain ischemia (surgically limiting blood flow to the front of the brain) results in a characteristic and rapid loss of neurons in the CA1 region of the hippocampus in gerbils (and other rodent species). Neurons are one of the most sensitive cells in the body to a short duration loss of oxygen. Sudden loss of oxygen (or blood flow) quickly kills or damages neurodn. Treating the injury after it happens (post treatment exposure) with HBO or HBA protected against neuronal loss in the CA1 (Figure 1). The NBO showed a statistically significant protection in the post-treatment exposure study [25] against the control group, but the effect was significantly smaller than either the HBO and HBA. Using an intervention prior to an injury (pre-conditioning, right columns) protected against the loss of neurons across all the exposure conditions (HBO, HBA and NBO), suggesting that oxidative signaling has robust mitochondrial and cellular protective effects [26]. This clearly demonstrates that HBA is not an inert control group, but a different oxygen dosing regimen. The notion that pressurized air is biologically inert is disproven prima facie (on its face or palm-to-face obvious) by this paper.
Figure 1 – Effects of hyperbaric oxygen (HBO), hyperbaric air (HBA) and normobaric oxygen (NBO) on neuronal survival in the CA1 region of the hippocampus in an ischemic model in gerbils. Columns are the average of 5 animals per exposure. Malek et al, statistical analysis: F-test, a= 0.05; *P < 0.001, **P < 0.05. Gadzyk et al: One-way ANOVA: a= 0.05; *P < 0.001, **P < 0.01
Study designs that assume the use of pressurized room air as a sham/placebo as not having a therapeutic effect need to be reassessed and conclusions revisited, including all the meta-analyses that have been done on HBO clinical trials. Studies that use experimental designs omitting a pressurized air group, use a cross-over group, limit the time spent at pressure to less than 5 minutes (for a control exposure) or compare baseline to post-treatment measures are the current best standards for hyperbaric medicine clinical research. Any other approach contradicts the abundant evidence [24, 27, 28] against the use of pressurized air or pure oxygen as a placebo/sham comparator, as well as sound clinical decision making [29].
The Current State of Evidence
The American Academy of Neurology (AAoN) has established a set of criteria [30-32] to delineate the different levels of clinical evidence to support or recommend against a medical intervention. This places the burden of proof on the reproducibility and quality of the clinical research as primary for a clinical recommendation to use (or not use) a therapeutic treatment/intervention.
There are four different classes of AAoN evidence classifications, but we are only interested in the most stringent type of classification:
Class I. A randomized, controlled clinical trial of the intervention of interest with masked or objective outcome assessment, in a representative population. Relevant baseline characteristics are presented and substantially equivalent among treatment groups or there is appropriate statistical adjustment for differences.
The following are also required:
a. Concealed allocation
b. Primary outcome(s) clearly defined
c. Exclusion/inclusion criteria clearly defined
d. Adequate accounting for dropouts (with at least 80% of enrolled subjects completing the study) and crossovers with numbers sufficiently low to have minimal potential for bias
e. For noninferiority or equivalence trials claiming to prove efficacy for one or both drugs, the following are also required*
1. The standard treatment used in the study is substantially similar to that used in previous studies establishing efficacy of the standard treatment (e.g., for a drug, the mode of administration, dose, and dosage adjustments are similar to those previously shown to be effective).
2. The inclusion and exclusion criteria for patient selection and the outcomes of patients on the standard treatment are substantially equivalent to those of previous studies establishing efficacy of the standard treatment.
3. The interpretation of the results of the study is based on an observed-cases analysis.
As the evidence classes for clinical trials are defined (I-IV), a threshold number of evidence sets have been established to provide guidance for recommendations for clinical application:
A. Established as effective, ineffective, or harmful (or established as useful/predictive or not useful/predictive) for the given condition in the specified population. (Level A rating requires at least two consistent Class I studies.)
B. Probably effective, ineffective, or harmful (or probably useful/predictive or not useful/predictive) for the given condition in the specified population. (Level B rating requires at least one Class I study or two consistent Class II studies.)
C. Possibly effective, ineffective, or harmful (or possibly useful/predictive or not useful/predictive) for the given condition in the specified population. (Level C rating requires at least one Class II study or two consistent Class III studies.)
D. Data inadequate or conflicting; given current knowledge, treatment (test, predictor) is unproven.
The current published literature will demonstrate that an evidence threshold for using HBOT in mTBI/PPCS has been reached.
National Studies: United States of America
Of the 19 case reports and clinical trials (Table 1) we identified in the English written literature, seven meet the criteria of Class I evidence. In the case of mild-to-moderate traumatic brain injury (mTBI), post-concussion syndrome (PCS)/persistent post-concussion syndrome (PPCS) with or without post-traumatic stress disorder (PTSD), there are six Class I clinical trials using HBOT [6, 9, 13, 14, 16, 17] and five of those studies focus on an active duty military or Veteran populations. In all cases, the studies show statistically significant improvement from pre-treatment measures at post-treatment assessment in the PTSD Checklist-Military (PCL-M) and/or the Rivermeade Post-Concussion Questionnaire (RPQ-Total) or in validated Neurocognitive tests (NSI: neurocognitive symptoms index).
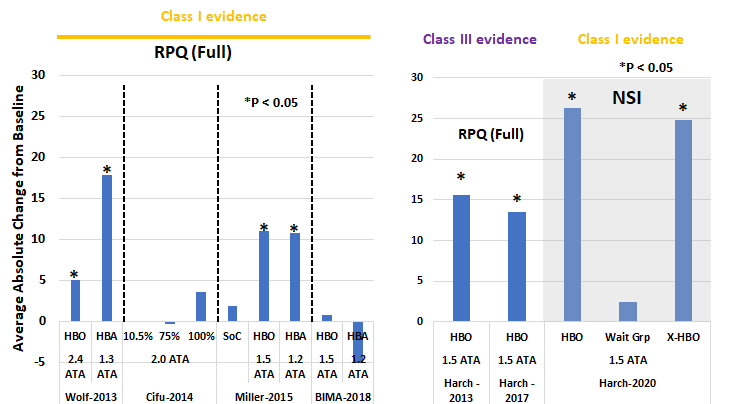
The results of Wolf et al, Cifu et al, the HOPPS study and the BIMA study (Figure 2, left) all show a significant improvement when looking at PTSD scores (using the PCL-M symptoms survey) for HBO and certain HBA groups. One study of note, the Miller et al clinical trial [13], had a standard-of-care (SoC) comparator group. This group, much like a waiting group in a cross-over trial, did not experience any significant improvements during the study period, while both treatment groups (HBO and HBA) experienced significant improvements. Claims of a placebo effect for the HBA groups as explanatory [39, 40] are not supported by the Cifu et al publication.
The outcome from the four DoD/VA studies are in agreement with the work carried out by Harch et al (Figure 2, right), with an improvement in symptoms scores that are in excess of what has been observed in non-HBOT clinical trials in the 2005-2015 time period [41]. One important parameter to point out in the Harch et al studies (2012, 2017 and 2020) [6, 18] [12] is the consistency between the three studies. Both earlier studies (2012 & 2017) were Pre-to-Post changes in symptom scores (Level III), while the 2020 study was a cross-over design (Level I). All of the HBOT exposure groups in Harch’s clinical trials experienced a significant improvement in their PTSD symptoms that were of 10 points or greater. The wait group in the Harch et al 2020 study reported no significant changes in PTSD scores in that time.
Figure 2 – Average absolute changes in post-traumatic stress disorder (PTSD) Checklist – Military (PCL-M) measures. Seven studies, four sponsored by the DoD/VA (left side) and three sponsored by private/public funding (right) display the average change from baseline (starting) measures to a 12-13-week measures after stopping HBOT, HBA or standard of care treatment. Stars designate statistically significant changes from starting (baseline) values, as reported by the authors. ATA, Atmosphere absolute; X-HBO, wait group crossed-over into HBO treatment; SoC, standard of care.
Improvements in the symptom’s assessment for the Rivermeade Post-Concussion Questionnaire (RPQ-Full) were also observed. Here the results from the DoD studies are mixed (Figure 3, left), with two studies reporting significant improvements and two studies showing non-significant changes. The study by Harch et al (Figure 3, right) shows improvement in RPQ values, while a later 2020 study, using a different scale (NSI) shows that the HBOT treatment group and the cross-over group have similar levels of improvement. The wait group showed no significant change. Placebo effects or Hawthorne effects were not noted.
Figure 3 – Average absolute changes in Rivermeade Post-concussion Symptoms Questionnaire (RPQ) measures for all 13 measures (Full) or Neurological Symptoms Index (NSI). Six studies, four sponsored by the DoD/VA (left side) and three sponsored by private/public funding (right) display the average change from baseline (starting) measures to a 12-13-week measures after stopping HBOT, HBA or standard of care treatment. Stars designate statistically significant changes from starting (baseline) values, as reported by the authors.
Recent Perspectives and Analysis
Of the six Class I studies (Table 1), some of the authors conclusions are not as clear cut and equivocally negative to HBOT as has been presented in the mainstream media reports [33-35], and editorials [36]. At the time of the GAO report (in 2015), active debate and calls for continued testing were being made and more clinical trials had yet to be published. The Government Accountability Office (GAO) [38] issued a balanced report of the field in 2015 on the overall effect of HBOT on mTBI/PCS/PPCS in a military population from the DoD/VA studies. A VA Evidence Brief [37] fell along the same lines as the GAO report and noted that further study was warranted and needed.
A recent review of the pooled analysis of the DoD/VA studies by Hart et al. [43] concluded the following:
“Trends from this pooled analysis suggest that there is some evidence to support continued evaluation of HBO2 as an intervention for mTBI and PCS, particularly given that those with mTBI-associated PCS and concomitant PTSD appeared to be more responsive to HBO2 than those with mTBI-associated PCS alone. While this evidence is not conclusive, given the lack of other competing standard-of-care treatments, further study of HBO2 in mTBI and PCS is worth consideration. Overall evidence from this analysis suggests a potential HBO2 dose-response relationship for PCS and PTSD outcomes. An appropriately controlled, definitive clinical trial of HBO2 for PTSD and PCS after mTBI should be considered. Additional work may be required to identify the optimal HBO2 dose for both PCS and PTSD.”
An analysis, comparing the outcomes from the HOPPS and BIMA studies by Weaver et al. [44] concluded that weighted composite total scores (post hoc measures) maximized the improvement seen in HBOT scores. Furthermore, it was observed in Weaver’s 2018 analysis [44] that
“The magnitude of improvement in self-reported assessments by 13 weeks is larger than any non-hyperbaric intervention previously reported… ” [9] and the observed reduction in improvements at 6 and 12 months “…could be due to an insufficient number of HBO2 sessions (i.e., underdosing)” [9].
Questions remain as to the type of population that will receive maximum benefit, dosing, frequency of treatment and best measures to use for an analysis of this therapy, yet a signal is detected in the noise of the data. These two most recent reviews by the thought leaders of the DoD/VA studies point to the fact that improvements with HBOT are present for those that are diagnosed with a mild-to-moderate TBI with or without PTSD (those with PTSD showed greater effects with HBOT); the ancillary questions, as to absolute efficacy, dosing, durability and identifying those that will show an absolute benefit from the intervention should be addressed in a separate study, without needing to wait for approval for its use in this population.
The inclusion of non-validated “sham/placebo” HBA has only served to confuse the outcomes of the HBOT groups. Although there is resistance and incredulity to the idea that low pressure air interventions could be therapeutic (and thus skew the overall interpretation of the HBOT clinical trials), the information supporting the HBA sham/placebo assertion is Class IV evidence (opinion). The evidence in support of HBA as having a therapeutic effect is at least Class II [24, 27, 28]. Until definitive evidence disproving pressurized air at or greater than 1.2 ATA has an effect on CNS injury models, defining HBA as a sham or placebo comparator is unproven, invalid and does not adhere to Popperian norms for hypothesis testing.
Hyperbaric oxygen therapy consistently improves symptoms and neurocognitive performance in military active duty and Veteran populations. Civilian populations treated with HBOT for their mTBI/PPCS show similar improvement, as will be discussed.
International Studies
A series of hyperbaric clinical studies in a civilian population [10, 11, 16, 45], published by an Israeli clinical group at the Sagol Center for Hyperbaric Medicine in the Assaf Harofeh Medical Center achieved similar results to the U.S. based studies. The study by Boussi-Gross et al [16] used a cross-over design, randomizing study subjects into an immediate treatment group and a wait-group (Figure 4). This provided a method to control for the participation effect (Hawthorne effect), as well as control for spontaneous recovery while waiting. Although the sets of computer-aided neurocognitive tests were not the same as the US DoD/VA studies, they show a similar parallel to the DoD/VA and Harch et al [6] study results. All study subjects improved to a statistically significant degree (immediate and cross-over group), while the wait group showed no significant changes during that time period. Indeed, the 2020 Harch et al clinical trial [6] replicated the Sagol Center [16] (Figure 2, right) publication in outcomes.
Figure 4 – Average absolute changes in Neuro Trax measures to a 12-13-week measures after stopping HBOT or wait period assessment. Non-significant changes from baseline values are designated by an arrow pointing down on the specific measure. All changes plotted in the graph are significant unless noted by an arrow.
A less discussed, but equally critical component of the studies is the cerebral blood flow analysis in the patient population in the Boussi-Gross et al [16] outcomes. Cerebral blood flow increased and remained at elevated levels post-HBOT treatment, when compared to baseline measures and to the Wait Group. These results are congruent with the SPECT outcomes from the Harch et al [18] study, which demonstrated that all study subjects had hypo-perfusion of blood flow in the brain prior to HBOT, but blood flow was elevated to levels seen in a healthy, non-injured population and stayed elevated after finishing their HBOT treatments.
Indeed, three more studies by Tal et al [11, 45] and Haddany et al [10] (two retrospective analyses of the patient population treated at the Sagol Center, one a prospective clinical trial) show clear improvements in neurocognitive performance (Figure 4). The studies included cerebral blood flow analysis and white matter tract density measures. The neurocognitive measures from the Tal and Haddany publications are consistent with the Boussi-Gross outcomes in their patient population. All four publications show clear increases in global cerebral blood flow and improved white matter density measures, indicating that white matter tracts are re-established in a mTBI population. This is a clear and reproducible physiological response that is sustained and long-lasting.
The published studies from Israel, using 1.5 and 2.0 ATA HBOT treatments, are consistent with the reported outcomes from the US DoD/VA sponsored studies and the Harch studies. Variability in the outcomes is not unexpected, but the consistency in symptoms and neurocognitive improvements between six independent clinical trial groups reduces the likelihood that this is a placebo effect or a methodological flaw inherent in the study designs. All the HBOT exposures in these civilian trials produced improvements (either at 1.5 or 2.0 ATA) and supported the results of the HBOT exposure group for the DoD/VA studies and the studies by Harch in Veteran study subjects. The consistency of outcomes between the Wolf, Miller, Harch, Weaver, Boussi-Gross and Tal studies indicates a replicable effect of HBOT across time, space and population base for a heterogenous central nervous system injury.
The Meaning of It All
How do we make sense of the large data set of HBOT treatments with mTBI study subjects, in terms of clinical results? Clearly, there are two camps of experts that are strongly promoting and strongly resistant to using HBOT for a neurological condition like mild-to-moderate traumatic brain injury. But, expert opinion is only that: opinion (a Class IV level of evidence under AAoN criteria). Currently, there are six Class I clinical trials. Obscuring the debate between these two camps is the HBA comparator used in the DoD/VA clinical trials. Setting aside the use of an unvalidated sham/placebo group [24, 27, 28] and just looking at the effects of HBOT against the starting baseline or against a standard of care/wait group, there are three studies (Miller [13], Boussi-Gross [16] and Harch [6]) that stand out and provide clear evidence of a therapeutic effect. These three Class I studies provide a clear and unambiguous evidence of an effect in neurocognitive improvements and symptom reduction in the HBOT treated group. By the AAoN criteria, this is a Level A recommendation for the use of HBOT in TBI/PPCS: Established as effective for the given condition in the specified population. (Level A rating requires at least two consistent Class I studies.)
An important consideration for the VA, DoD and private insurers to consider is the recommendations that have been set forth by the Agency for Healthcare Research and Quality (AHRQ, a unit in the Public Health Services of the USA) recommendations for future HBOT research for TBI [29]. One pertinent comment was the following:
“Whether placebo-controlled trials are necessary to evaluate HBOT has received a great deal of attention in discussions about HBOT. Participants on all sides of this debate make the assumption that an “evidence-based” approach implies devotion to double-blind, placebo-controlled trials without regard to practical or ethical considerations. This assumption is false. Double-blind, placebo-controlled trials are the “gold standard” for government regulators overseeing the approval of new pharmaceuticals, but not for clinical decision-making or insurance coverage decisions. Evidence-based clinical decisions rely more heavily on comparisons of one treatment to other potentially effective therapies, not to placebos [author bolding].”
HBOT as a therapy for TBI/PCS/PPCS has met the bar for inclusion as a regular intervention. In the United States there are no phase III clinical trials (past or current) that have tested any drug, therapy or device for efficacy on TBI/PCS/PPCS or PTSD treatments. Yet many drugs and therapies are routinely prescribed by both the VA and DoD for TBI/PCS/PPCS and PTSD, promoted to practitioners and recommended for use in several programs, without Phase III data. As long as a clinical improvement is observed, compared to a standard treatment offered in a clinical setting, then clinical decisions for the use of a therapy supersede other requirements. This has been amply documented and commented on by Hart et al [43]. HBOT is the only therapy that reports significant and large scale improvements in symptoms scores at 13 weeks post-therapy termination [44], exceeding any therapy currently used.
The issue of safety is a non-issue, when compared to other common procedures and therapies. Since 2008 the DoD and the VA [46] have noted that HBOT is a safe treatment. Serious or life-threatening adverse reactions are rare or almost non-existent, as all patients are given a full medical history prior to undergoing pressurization to identify the vulnerable population.
The use of lower pressure regimes (1.5-2.0 ATA) are not “standard” in the sense that pressures of 2.4-3.0 ATA are routinely used for skin flaps, non-healing ulcers, decompression illness and carbon-monoxide poisoning (just to name a few) in the hyperbaric medicine and wound treatment fields. These lower pressure regimes (1.5-2.0 ATA) are considered off-label under the covered indications for Centers for Medicare and Medicaid Services (CMS) guidelines, which the VA follows. CMS has, as a policy, the obligation of practicing evidence-based medicine. In this case, evidence is present to make a change in CMS and VA policy to include HBOT for mTBI as a covered indication. To quote Dr. George Wolf, in his 2015 review [47]:
““Placebo effect in our previous reports has been considered as why there was no significant statistical difference in this study...However, both groups showed improvement in scores and thus a benefit. Given the studies demonstrating hydrostatic pressure effects and results of Boussi-Gross’ crossover study, our design could be considered a treatment comparison vs. a true sham with a therapeutic effect from both increased oxygen partial pressure and hydrostatic pressure. A Type II statistical error cannot be ruled out...There is a potential gain and no potential loss. The VA/Clinical Practice Guidelines define a “B evidence rating” as “a recommendation that clinicians provide (the service) to eligible patients”. At least fair evidence was found that the intervention improves health outcomes and concludes that benefits outweigh harm””
Several authors have opposed the use of HBOT for TBI citing cost, time commitment for treatment, limited locations for service and lack of a phase III clinical trial to establish efficacy and specificity to treat a specific clinical diagnosis [36, 41, 48, 49]. The cited concerns are secondary to the reported and clinically relevant effects on symptoms and neurocognitive improvements. The objections are not supported by CMS or VA/Clinical Practice Guidelines. Informed consent for all available treatment options, even those deemed too costly or too difficult by the physician, are not the sole purview of the physician. Patients ultimately make the decision to use or decline a particular intervention, but only after being provided all options to make an informed medical decision. There is abundant clinical evidence for use of HBOT in individuals diagnosed with mTBI with or without PTSD. HBOT is an effective intervention that can achieve large-scale reduction in symptoms, especially PTSD, as well as recover cognitive performance.
The issue facing the medical community, medical armed service and the Veterans Administration is a cultural and political battle. More than twelve years have been spent by the combined agencies and medical bodies asserting that HBOT does not work, yet the current efforts by the same agencies are not denting the current suicide epidemic and opioid addiction. The DoD and VA have spent at least $9.2 billion since 2010 attempting to treat PTSD and TBI injuries and the ever-increasing suicide epidemic linked with this condition in our nations Veterans [50]. Drug interventions are inadequate for TBI and PTSD recovery [51-53] and cases of prescribed medication have been linked to Veteran suicide [54, 55] . Enough time has elapsed for the evidence to come in (it is here and abundant) and it is incumbent for the VA and the DoD to stop the second coming of Agent Orange 2.0. Former Secretary of the Army Martin Hoffman (Col., Army, Ret.) [56] labeled the inability of the DoD and VA to address the TBI/PTSD epidemic as being in the same vein as the Agent Orange legal battle: failure to address service connected injuries will ensure that veterans do not receive treatment and limp along in their post-service lives.
Final Words…
It is time to recognize the clinical utility of this treatment in an mTBI/PCS/PPCS setting. The ability to improve the lives of millions of brain-wounded and avoid lifetimes of lost earnings and the social impact of avoidable suffering is real and achievable. The evidence threshold has been achieved with this intervention and it is incumbent upon Centers for Medicare and Medicaid (DHHS), VA and private insurers to cover this intervention. Critical for this to occur is the role of the physician in understanding and applying this intervention on behalf of their patients. Improvement and recovery is possible for the majority of brain injured patients with HBOT and the potential to achieve synergies in combination with other interventions is possible.
And by other interventions, I am looking at the integrated approaches that were developed in places like Rocky Mountain Hyperbarics. Dr. Pepe Ramirez (SgtMaj, USMC, ret.) helped to develop a coordinated and integrated approach, using HBOT and psychotherapy for veterans with TBI and PTSD. Lots of good results were achieved - +300 service members treated - with little to no help from the US government and mostly privately funded.
Thank you Caleb Gates, Grady Birdsong (Cpl, USMC, ret.), Bob Fischer (Col, USMC, ret.), Wayne Quick, Ray Cralle (Pvt, USMC, ret.), Ryan Fullmer, Eddie Gomez, Rob Beckman, Ph.D. (Cpt, USAF, ret.), Eric Koleda (USAF, ret.), James Bauerle (BGen, Army, ret.), James Wright (Col, USAF, ret.), Thomas Fox (Maj, Army, ret.), Paul Hoffecker, Dr. Denise Dunham, Dr. Carol Hedrick, Dr. Henry Ealy and I could go on… too many have given without recognition and at great personal cost.
‘Nuff said…for now.
References
1. Cameron, H.A. and R. McKay, Stem cells and neurogenesis in the adult brain. Curr Opin Neurobiol, 1998. 8(5): p. 677-80.
2. Alvarez-Buylla, A. and C. Lois, Neuronal stem cells in the brain of adult vertebrates. Stem Cells, 1995. 13(3): p. 263-72.
3. Xie, F., H. Liu, and Y. Liu, Adult Neurogenesis Following Ischemic Stroke and Implications for Cell-Based Therapeutic Approaches. World Neurosurg, 2020. 138: p. 474-480.
4. Matsuda, T. and K. Nakashima, Natural and forced neurogenesis in the adult brain: Mechanisms and their possible application to treat neurological disorders. Neurosci Res, 2020.
5. Bobkova, N.V., et al., Neuroregeneration: Regulation in Neurodegenerative Diseases and Aging. Biochemistry (Mosc), 2020. 85(Suppl 1): p. S108-S130.
6. Harch, P.G., et al., Hyperbaric oxygen therapy for mild traumatic brain injury persistent postconcussion syndrome: a randomized controlled trial. Med Gas Res, 2020. 10(1): p. 8-20.
7. Shytle, R.D., et al., Retrospective Case Series of Traumatic Brain Injury and Post-Traumatic Stress Disorder Treated with Hyperbaric Oxygen Therapy. Cell Transplant, 2019. 28(7): p. 885-892.
8. Mozayeni, B.R., et al., The National Brain Injury Rescue and Rehabilitation Study - a multicenter observational study of hyperbaric oxygen for mild traumatic brain injury with post-concussive symptoms. Med Gas Res, 2019. 9(1): p. 1-12.
9. Weaver, L.K., et al., Hyperbaric oxygen for post-concussive symptoms in United States military service members: a randomized clinical trial. Undersea Hyperb Med, 2018. 45(2): p. 129-156.
10. Hadanny, A., et al., Effect of hyperbaric oxygen therapy on chronic neurocognitive deficits of post-traumatic brain injury patients: retrospective analysis. BMJ Open, 2018. 8(9): p. e023387.
11. Tal, S., et al., Hyperbaric Oxygen Therapy Can Induce Angiogenesis and Regeneration of Nerve Fibers in Traumatic Brain Injury Patients. Front Hum Neurosci, 2017. 11: p. 508.
12. Harch, P.G., et al., Case control study: hyperbaric oxygen treatment of mild traumatic brain injury persistent post-concussion syndrome and post-traumatic stress disorder. Med Gas Res, 2017. 7(3): p. 156-174.
13. Miller, R.S., et al., Effects of hyperbaric oxygen on symptoms and quality of life among service members with persistent postconcussion symptoms: a randomized clinical trial. JAMA Intern Med, 2015. 175(1): p. 43-52.
14. Cifu, D.X., et al., Hyperbaric oxygen for blast-related postconcussion syndrome: three-month outcomes. Ann Neurol, 2014. 75(2): p. 277-86.
15. Cifu, D.X., et al., The effect of hyperbaric oxygen on persistent postconcussion symptoms. J Head Trauma Rehabil, 2014. 29(1): p. 11-20.
16. Boussi-Gross, R., et al., Hyperbaric oxygen therapy can improve post concussion syndrome years after mild traumatic brain injury - randomized prospective trial. PLoS One, 2013. 8(11): p. e79995.
17. Wolf, G., et al., The effect of hyperbaric oxygen on symptoms after mild traumatic brain injury. J Neurotrauma, 2012. 29(17): p. 2606-12.
18. Harch, P.G., et al., A phase I study of low-pressure hyperbaric oxygen therapy for blast-induced post-concussion syndrome and post-traumatic stress disorder. J Neurotrauma, 2012. 29(1): p. 168-85.
19. Stoller, K.P., Hyperbaric oxygen therapy (1.5 ATA) in treating sports related TBI/CTE: two case reports. Med Gas Res, 2011. 1(1): p. 17.
20. Wright, J.K., et al., Case report: Treatment of mild traumatic brain injury with hyperbaric oxygen. Undersea Hyperb Med, 2009. 36(6): p. 391-9.
21. Harch, P.G., et al., Low pressure hyperbaric oxygen therapy and SPECT brain imaging in the treatment of blast-induced chronic traumatic brain injury (post-concussion syndrome) and post traumatic stress disorder: a case report. Cases J, 2009. 2: p. 6538.
22. Lin, J.W., et al., Effect of hyperbaric oxygen on patients with traumatic brain injury. Acta Neurochir Suppl, 2008. 101: p. 145-9.
23. Hardy, P., et al., Pilot case study of the therapeutic potential of hyperbaric oxygen therapy on chronic brain injury. J Neurol Sci, 2007. 253(1-2): p. 94-105.
24. Harch, P.G., Hyperbaric oxygen in chronic traumatic brain injury: oxygen, pressure, and gene therapy. Med Gas Res, 2015. 5: p. 9.
25. Malek, M., et al., Hyperbaric oxygen and hyperbaric air treatment result in comparable neuronal death reduction and improved behavioral outcome after transient forebrain ischemia in the gerbil. Exp Brain Res, 2013. 224(1): p. 1-14.
26. Gamdzyk, M., et al., Hyperbaric oxygen and hyperbaric air preconditioning induces ischemic tolerance to transient forebrain ischemia in the gerbil. Brain Res, 2016. 1648(Pt A): p. 257-265.
27. Figueroa, X.A. and J.K. Wright, Hyperbaric oxygen: B-level evidence in mild traumatic brain injury clinical trials. Neurology, 2016. 87(13): p. 1400-6.
28. Figueroa, X.A. and J.K. Wright, Clinical results in brain injury trials using HBO2 therapy: Another perspective. Undersea Hyperb Med, 2015. 42(4): p. 333-51.
29. McDonagh, M., et al., Hyperbaric oxygen therapy for brain injury, cerebral palsy, and stroke. Evid Rep Technol Assess (Summ), 2003(85): p. 1-6.
30. Gross, R.A. and K.C. Johnston, Levels of evidence: Taking Neurology to the next level. Neurology, 2009. 72(1): p. 8-10.
31. Gronseth, G. and J. French, Practice parameters and technology assessments: what they are, what they are not, and why you should care. Neurology, 2008. 71(20): p. 1639-43.
32. French, J. and G. Gronseth, Lost in a jungle of evidence: we need a compass. Neurology, 2008. 71(20): p. 1634-8.
33. Zorota, G., Hyperbaric chamber treatments did not help with mild TBI, in USA Today. 2013: On-line.
34. Kohn, d., Could hyperbaric treatment heal the brain?, in The Washington Post. 2018: Online.
35. Mieir, B. and D. Ivoiry, Effective Concussion Treatment Remains Frustratingly Elusive, Despite a Booming Industry, in The New York Times. 2015.
36. Bennett, M.H., Evidence brief: hyperbaric oxygen therapy (HBOT) for traumatic brain injury and/or post-traumatic stress disorder. Diving Hyperb Med, 2018. 48(2): p. 115.
37. Peterson, K., et al., Evidence Brief: Hyperbaric Oxygen Therapy (HBOT) for Traumatic Brain Injury and/or Post-traumatic Stress Disorder, in VA Evidence Synthesis Program Evidence Briefs. 2018: Washington (DC).
38. Draper, D.A., DEFENSE HEALTH CARE - - Research on Hyperbaric OxygenTherapy to Treat Traumatic Brain Injury and Post-Traumatic Stress Disorder, U.S.G.A. Office, Editor. 2015.
39. Bennett, M.H., Hyperbaric medicine and the placebo effect. Diving Hyperb Med, 2014. 44(4): p. 235-40.
40. Hoge, C.W. and W.B. Jonas, The ritual of hyperbaric oxygen and lessons for the treatment of persistent postconcussion symptoms in military personnel. JAMA Intern Med, 2015. 175(1): p. 53-4.
41. Wilson, S.H., et al., Review of recent non-hyperbaric oxygen interventions for mild traumatic brain injury. Undersea Hyperb Med, 2016. 43(5): p. 615-627.
42. Using the PTSD Checklist for DSM-5 (PCL-5), N.C.f.P.P.S. Disorder, Editor., www.ptsd.va.gov.
43. Hart, B.B., et al., Hyperbaric oxygen for mTBI-associated PCS and PTSD: Pooled analysis of results from Department of Defense and other published studies. Undersea Hyperb Med, 2019. 46(3): p. 353-383.
44. Weaver, L.K., et al., A composite outcome for mild traumatic brain injury in trials of hyperbaric oxygen. Undersea Hyperb Med, 2019. 46(3): p. 341-352.
45. Tal, S., et al., Hyperbaric oxygen may induce angiogenesis in patients suffering from prolonged post-concussion syndrome due to traumatic brain injury. Restor Neurol Neurosci, 2015. 33(6): p. 943-51.
46. Harch, P.G., TESTIMONY FOR THE RECORD: Suicides in U.S. Military Personnel, Veterans of the War in Iraq and Afghanistan, and the Core Medical Treatment for Mild-Moderate Traumatic Brain Injury & PTSD, TreatNOW.org, Editor. 2010, International Hyperbaric Medical Association: International Hyperbaric Medical Association 8210 Cinder Bed Road, Ste C-3, Lorton, Virginia 22079-1135. p. 20.
47. Wolf, E.G., et al., Cognitive function in a traumatic brain injury hyperbaric oxygen randomized trial. Undersea Hyperb Med, 2015. 42(4): p. 313-32.
48. Crawford, C., et al., Is Hyperbaric Oxygen Therapy Effective for Traumatic Brain Injury? A Rapid Evidence Assessment of the Literature and Recommendations for the Field. J Head Trauma Rehabil, 2017. 32(3): p. E27-E37.
49. Mitchell, S.J. and M.H. Bennett, Unestablished indications for hyperbaric oxygen therapy. Diving Hyperb Med, 2014. 44(4): p. 228-34.
50. in Treatment for Posttraumatic Stress Disorder in Military and Veteran Populations: Final Assessment. 2014: Washington (DC).
51. Lee, E.A., Complex contribution of combat-related post-traumatic stress disorder to veteran suicide: facing an increasing challenge. Perspect Psychiatr Care, 2012. 48(2): p. 108-15.
52. Smith, E.G., et al., Treatment of Veterans with depression who died by suicide: timing and quality of care at last Veterans Health Administration visit. J Clin Psychiatry, 2011. 72(5): p. 622-9.
53. Nierenberg, A.A., The Congressional antidepressant hearings: exploring the relationship between medication and veteran suicide. CNS Spectr, 2010. 15(6): p. 348-9.
54. Flock, E., Medical Reports, Veterans' Families Suggest VA Is Overmedicating Vets. US News and World Report, 2013.
55. Reno, J., “Medicating Our Troops Into Oblivion”: Prescription Drugs Said To Be Endangering U.S. Soldiers. International Business Times, 2014.
56. Figueroa, X.A., Sacred Duty, Broken Promise: A Call to Action in Treating Our Wounded. Combat Stress, 2018. 7(1): p. 8.